Allogeneic hematopoietic cell transplantation (HCT) is the only curative treatment for a broad range of hematological malignancies. Unfortunately, the current standard of care relies on untargeted toxic conditioning regimens associated with severe morbidity and mortality curtailing HCT's wider use. In the last decade, antigen-specific cell depleting therapies have revolutionized clinical practice in hematology. Although target selection remains complex, the pan-hematopoietic marker CD45, a protein tyrosine phosphatase which is exclusively expressed on all nucleated hematopoietic cells, could enable targeted depletion of the entire hematopoietic system including HSCs. Combined with HSCs engineered to be shielded from a CD45-targeting antibody-drug conjugate (ADC), this approach could create a near-universal strategy to replace a diseased hematopoietic system, irrespective of disease etiology or cell type.
We performed alanine (Ala) scanning of the entire extracellular domain of CD45 to identify specific residues forming the epitopes for three different monoclonal antibodies (mAbs). Next, we screened base editors (BE) to generate amino acid substitutions that reduced or abolished the binding of specific mAbs, purified the first two extracellular regions of the most promising protein variants (CD45 D1-D2) and measured their binding to the mAbs by biolayer interferometry (BLI). Two antibodies showed drastically reduced or no binding to some of the CD45 variants. Some variants only reduced the binding of the mAb of the corresponding epitope while others affected the binding of mAbs from multiple epitopes located in more distant regions. These results suggested that several CD45 amino acid exchanges altered the receptor structure in a way that spanned at least two structural domains. This interpretation is supported by a substantial decrease in protein thermal stability, calling for caution when selecting suitable variants. Nevertheless, through a series of experiments and optimizations of the sgRNAs we identified a BE (ABE8e-SpRY + sgRNA49.3) that resulted in biophysically stable protein variants with editing of about 50% of hematopoietic stem and progenitor cells (HSPCs). These base edited HSPCs formed equal numbers of colonies as well as relative numbers of myeloid and erythroid colonies comparable to control HSPCs. Furthermore, they engrafted and supported multi-lineage cell differentiation in NBSGW host mice comparable to non-edited control HSPCs. Secondary transplantation experiments confirmed successful editing of long-term reconstituting HSCs (LT-HSCs). Collectively, these results indicate that the edited HSPCs were functionally intact.
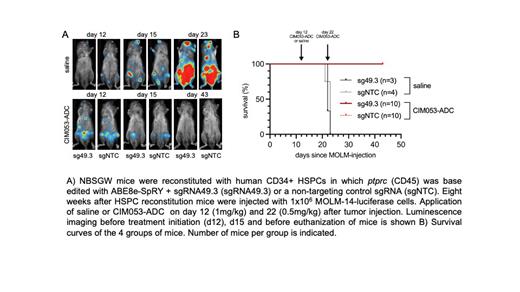
We further developed a humanized, Fc silent CD45-ADC designated CIM053-ADC. A single dose of 0.5mg/kg was very well tolerated by NBSGW mice engrafted with hCD34 + HSPCs and effectively depleted human hematopoietic cells including LT-HSCs. Next, we injected luciferase marked MOLM-14 cells, an aggressive myeloid tumor cell line, in mice priorly xenografted with edited or control human HSPCs. CIM053-ADC treatment was initiated 12 days after tumor injection when mice demonstrated clearly detectable, extensive and multilocular disease ( Fig. A). We deliberately assigned the mice with the most advanced disease to the ADC group. Mice then obtained saline or two doses of CIM053-ADC i.v.. Tumors were cleared in all CIM053-ADC treated mice whereas in all the mice that obtained saline the MOLM-14 cells rapidly grew and mice had to be euthanized 21-23 days post tumor injection ( Fig. B). Importantly, CIM053-ADC eliminated unedited hCD45 + non-tumor cells but base edited cells persisted in all tested organs.
In summary, we developed a novel, highly potent tumor-selective therapy that spares a shielded hematopoietic system. Since CIM053-ADC targets the pan-hematopoietic marker CD45 this approach may not only be applicable to treat AML but most hematologic malignancies and possibly other diseases requiring HCT.
Disclosures
Lepore:Cimeio Therapeutics AG: Current equity holder in private company, Ended employment in the past 24 months, Patents & Royalties. Simonetta:Incyte: Consultancy; Kite/Gilead: Consultancy; Kite/Gilead: Other: speaker fees; Incyte: Other: speaker fees; Kite/Gilead: Other: Travel support; Novartis: Other: Travel support; BMS/Celgene: Consultancy; Astra Zeneca: Other: Travel support; Neovii: Other: Travel support; Janssen: Other: Travel support; BMS/Celgene: Research Funding; Novartis: Research Funding; Kite/Gilead: Research Funding. Camus:Cimeio Therapeutics AG: Current Employment, Current equity holder in private company, Patents & Royalties. Durzynska:Cimeio Therapeutics: Current equity holder in private company, Patents & Royalties. Kirby:ADC Therapeutics: Current Employment, Current equity holder in publicly-traded company. Van Berkel:ADC Therapeutics: Current Employment, Current equity holder in publicly-traded company, Patents & Royalties. Kunz:Cimeio Therapeutics: Current equity holder in private company. Urlinger:Cimeio Therapeutics AG: Current Employment, Current equity holder in private company, Patents & Royalties. Jeker:Cimeio Therapeutics AG: Consultancy, Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees, Patents & Royalties, Research Funding.